A Modular Dual-Network Anhydrous Matrix as a Platform for Multi-Mechanistic Antimicrobial Delivery
Expanding Beyond Single-Active Systems
Persistent microbial infections are increasingly understood as problems of microenvironment and structure rather than simple failures of antimicrobial potency. Biofilms, lipid-rich niches, and reduced diffusion gradients protect microorganisms from conventional aqueous formulations.
We describe a modular, dual-network, anhydrous matrix platform designed to host and control a wide spectrum of active chemistries. The system consists of a polarity-separated architecture incorporating a hydrophilic glycerine domain and a hydrophobic oil domain, structured within a thixotropic framework. This configuration allows integration of polar, lipophilic, water-soluble, alcohol-soluble, and encapsulated actives without compromising stability.
Rather than focusing on a single antimicrobial agent, the platform emphasizes kinetic control, diffusion modulation, and sequential stress induction as primary design principles.
1. Introduction: The Microenvironment Problem
Antimicrobial resistance is often framed as a genetic arms race. However, resistance in chronic infections frequently arises from physical and structural barriers such as biofilms. These extracellular polymeric matrices reduce drug penetration, alter metabolic states, and create protected micro-niches.
Traditional topical formulations are predominantly water-based. While effective for delivering hydrophilic compounds, they do little to alter the structural environment of established microbial communities. In some cases, they may even sustain local hydration conditions favorable to microbial survival.
A materials-first approach reframes the problem:
Control water activity.
Control diffusion.
Control polarity.
Control release kinetics.
This perspective shifts innovation from discovering stronger molecules to engineering more intelligent delivery systems.
2. Platform Architecture
2.1 Anhydrous Continuous Phase
The absence of bulk water is a foundational design feature.
Benefits include:
Reduced hydrolytic degradation of actives
Improved stability of redox intermediates
Lower microbial contamination risk within the formulation
Controlled activation at the tissue interface
Importantly, anhydrous does not mean incompatible with water-soluble actives. It means activation is regulated rather than spontaneous.
2.2 Dual-Network Polarity Separation
The matrix consists of two interpenetrating domains:
Hydrophilic Domain (Glycerine-Based)
Supports polar actives
Enables moisture-triggered activation
Suitable for oxidative systems, enzymes, peptides, and small polar molecules
Hydrophobic Domain (Oil-Based)
Supports lipophilic actives
Enables diffusion into lipid-rich tissues (sebum, nail bed, intercellular lipids)
Suitable for lipophilic antifungals, radical donors, terpene systems, and photosensitizers
The physical separation of domains prevents premature cross-reaction and allows independent kinetic tuning.
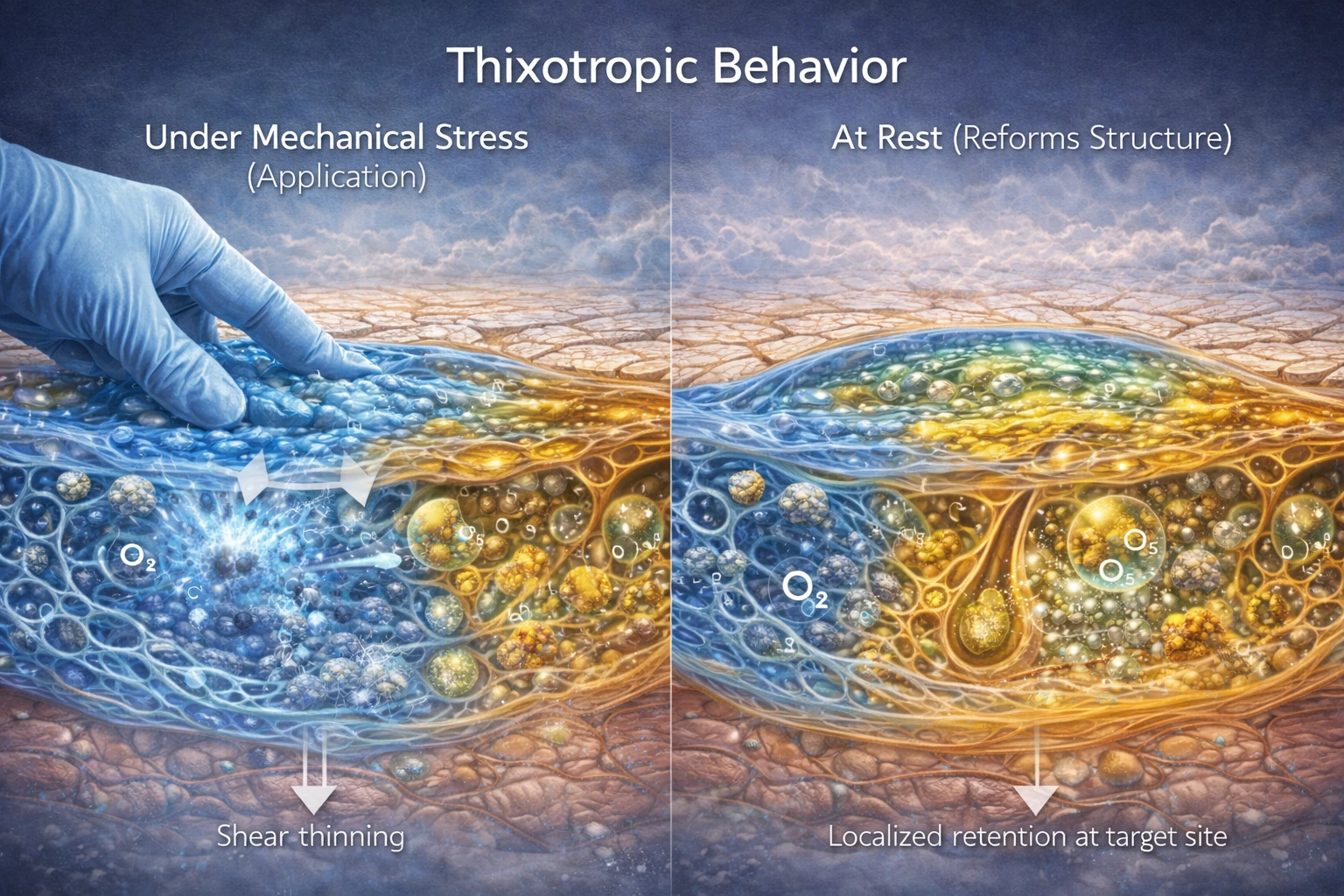
2.3 Thixotropic Structural Behavior
The matrix exhibits shear-thinning properties, meaning:
It flows under mechanical stress (application).
It reforms structure at rest.
This ensures localized retention at the target site and prolongs exposure time without uncontrolled spreading.
Residence time is as important as potency.
3. Expansion of Active Classes
The modular design allows integration of diverse actives. Below we outline key categories under investigation.
3.1 Oxidative and Redox Systems
Oxidative stress remains one of the most universal antimicrobial strategies because it targets fundamental cellular structures such as membranes and proteins.
Potential systems include:
Stabilized hydrogen peroxide complexes
Peroxymonocarbonate systems
Organic peresters
Lipid-phase radical donors
Ozone-modified fatty acid derivatives
By adjusting polarity placement, burst intensity and sustained release phases can be independently controlled.
3.2 Gasotransmitter Systems
Nitric oxide (NO) represents an intriguing antimicrobial and biofilm-disruptive molecule.
Controlled NO donors may:
Interfere with quorum sensing
Promote biofilm dispersal
Induce localized antimicrobial stress
Modulate host immune response
Encapsulation strategies can allow slow gas release within a structurally confined matrix.
Hydrogen sulfide and other gasotransmitters remain exploratory but mechanistically interesting.
3.3 Biofilm Matrix Destabilization
Sequential stress induction is a strategic focus.
Before applying strong oxidative or lipophilic stressors, structural destabilization may improve penetration.
Under evaluation:
EDTA and metal chelation systems
N-acetylcysteine for disulfide bond disruption
D-amino acids that influence biofilm dispersal
DNase and enzyme-based EPS degradation
These systems do not kill directly. They modify structure to enhance susceptibility.

3.4 Lipid-Targeted Antifungal Modules
Fungal infections often reside within keratinized or lipid-rich tissues.
Lipophilic actives of interest include:
Undecylenic acid
Chitosan derivatives
Farnesol
Terpene fractions (e.g., thymol, carvacrol)
Quinone derivatives
Oil phase selection determines penetration profile and release kinetics.
3.5 Enzyme Encapsulation
Enzymes require stabilization within micro-reservoirs to maintain activity in an anhydrous environment.
Encapsulation strategies under consideration include:
Microaqueous droplets
Polymer-coated particles
Lipid vesicles
Porous carrier adsorption
Enzyme systems enable targeted structural disruption of microbial extracellular matrices.
3.6 Photodynamic Activation
Photosensitizers represent a mechanism of temporal control.
Compounds such as porphyrins and methylene blue derivatives generate reactive oxygen species only upon light activation.
This introduces:
Spatial control (localized placement)
Temporal control (activation on demand)
The matrix localizes the sensitizer. Light provides the trigger.

4. Encapsulation as a Compatibility Tool
A major advantage of this platform is the ability to incorporate actives traditionally considered incompatible with anhydrous systems.
Water-soluble or alcohol-soluble actives can be:
Microencapsulated in aqueous cores
Dispersed in stabilized microdroplets
Immobilized onto structured carriers
This allows integration of antibiotics, enzymes, peptides, and other polar molecules without destabilizing the overall architecture.
Encapsulation transforms solubility limitations into engineering variables.
5. Research Considerations
Each active requires systematic evaluation:
Chemical compatibility testing
Rheological analysis
Stability under temperature variation
Cytotoxicity assessment
Diffusion modeling
Biofilm disruption assays
No single mechanism should be assumed sufficient across all indications. The platform enables combinatorial experimentation.
6. Discussion
The central innovation lies not in a specific antimicrobial compound but in the structured control of reaction environments.
By separating polarity domains and enabling encapsulation, the matrix becomes a modular reaction ecosystem.
This architecture allows:
Sequential stress application
Burst and sustained release phases
Tissue-specific oil selection
Reduced premature degradation
Adaptive integration of future chemistries
Instead of designing products around a molecule, we design systems around controlled microenvironment engineering.
7. Conclusion
The dual-network anhydrous matrix is best understood as a research platform rather than a single therapeutic product.
Its value lies in:
Polarity control
Encapsulation flexibility
Diffusion tuning
Thixotropic localization
Modular active integration
Future development will focus on pairing destabilization agents with oxidative or lipid-phase stressors, followed by tissue-supportive co-actives.